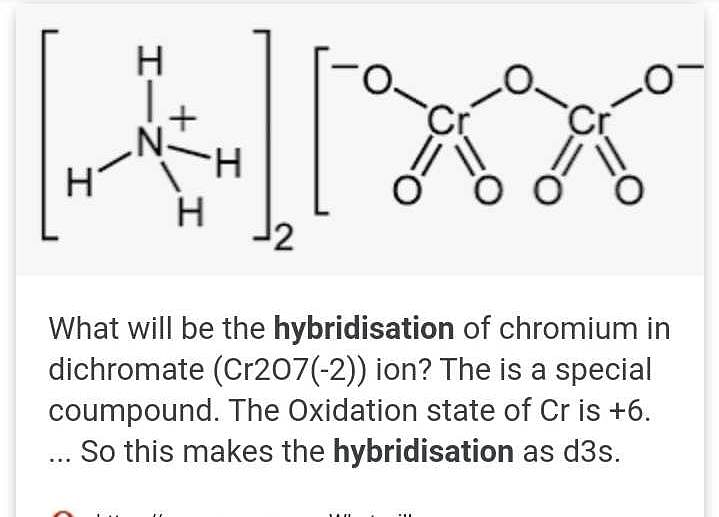
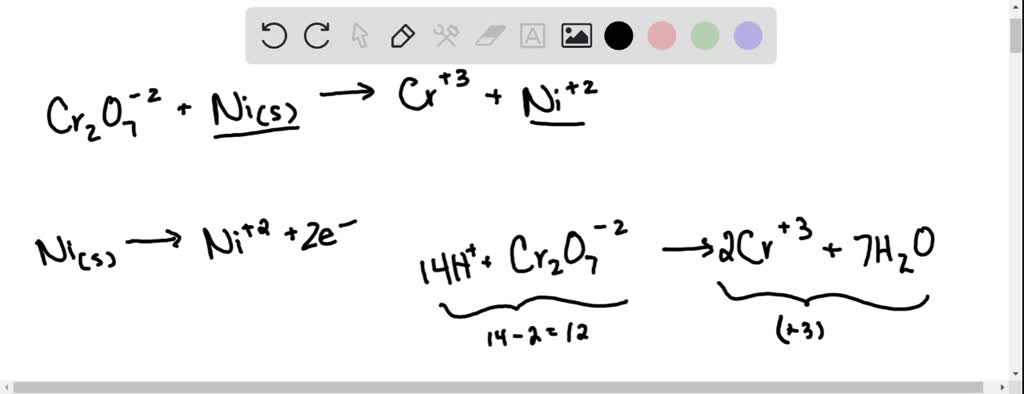
SOLVED: Balance the following reactions in an acidic solution (not all will need Ht ions) Cr (s) + Ni2+ 7 Cr 3+ (aq) + Ni (s) 2. Clz (aq) + Br 7

Chromium transition metal Chemistry chromium(III) Cr3+ complex ions chromate(VI) CrO42- dichromate(VI)Cr2O72- redox chemical reactions principal +3 +6 oxidation states ligand substitution GCE AS A2 IB A level inorganic chemistry revision notes

Calculate the equilibrium constant for the reaction: 3Sn(s) + 2Cr2O7^2 - + 28 H^+→ 3 Sn^+4 + 4Cr^3 + + 14H2O E^∘ for Sn/Sn^2 + = 0.136 V E^∘ for Sn^2 + /

IB chemistry online - Transition elements-Colors: From left to right: Ti+2, V+3, VO2+, Cr+3, Cr2O7-2, Mn+2, MnO4-1, Fe+3, Co+2, Ni+2, Cu+2 | Facebook

Palladium-Catalyzed Cycloisomerization of 2-Ethynylbiaryls to 9-Methylidene Fluorenes | Organic Letters

Write the name and formula of the chemical reagent used for the diagnosis of Ni^2+ ion in a solution. | Homework.Study.com

SOLVED: In this experiment you will balance several oxidation-reduction equations using the half-reaction method. 1.MnO4-1 (aq) + Cl-1 (aq) -> Mn+2 (aq) + Cl2 (g) (acidic solution) 2.H2S (aq)+ Cr2O7-2 (aq) ->

SOLVED: Balance the following reactions in an acidic solution (not all will need Ht ions) 1. Cr (s) + Ni2+ 7 Cr % (aq) + Ni (s) 2. Clz (aq) + Br

The coordination sphere around the nickel(II) atom in the structure of... | Download Scientific Diagram

Scheme 1. Schematic illustration of the formation of Ag-Ni core-shell... | Download Scientific Diagram
Color of Transition Metal Complexes The variety of color among transition metal complexes has long fascinated the chemists. For
Color of Transition Metal Complexes The variety of color among transition metal complexes has long fascinated the chemists. For
![Polar Compounds Constructed with the [Cr2O7]2- Anion | Inorganic Chemistry Polar Compounds Constructed with the [Cr2O7]2- Anion | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/ic000953y/asset/images/large/ic000953yn00001.jpeg)

![PDF] Polar compounds constructed with the [Cr2O7]2- anion. | Semantic Scholar PDF] Polar compounds constructed with the [Cr2O7]2- anion. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/01bd3ffb43425676c7723ca53225f700e4d30c92/2-Table3-1.png)

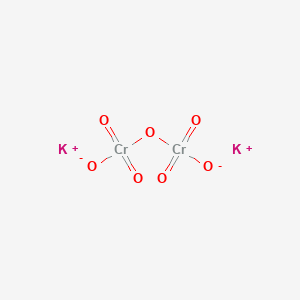
![Cr2O7]2- - Dichromate Cr2O7]2- - Dichromate](http://www.chemtube3d.com/images/gallery/PNGfiles%20structures/I619STB1.png)