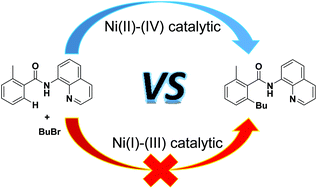
Ni(i)–Ni(iii) vs. Ni(ii)–Ni(iv): mechanistic study of Ni-catalyzed alkylation of benzamides with alkyl halides - Organic Chemistry Frontiers (RSC Publishing)

Suzuki-Miyaura coupling catalyzed by a Ni(II) PNP pincer complex: Scope and mechanistic insights - ScienceDirect

Mechanisms of Nickel-Catalyzed Coupling Reactions and Applications in Alkene Functionalization | Accounts of Chemical Research

Nickel-catalysed C-H arylation. a Ni(II)-catalysed C-H bond arylation... | Download Scientific Diagram
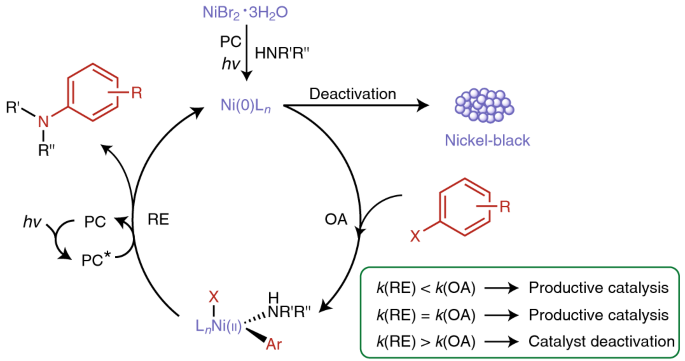
Overcoming limitations in dual photoredox/nickel-catalysed C–N cross-couplings due to catalyst deactivation | Nature Catalysis

Mechanisms of Nickel-Catalyzed Coupling Reactions and Applications in Alkene Functionalization | Accounts of Chemical Research

Figure 2 from The mechanism of directed Ni(ii)-catalyzed C–H iodination with molecular iodine† †Electronic supplementary information (ESI) available: Additional geometrical information and free energy surfaces for the examined reaction pathways and ...

Ligand-Enabled Ni(II)-Catalyzed Hydroxylarylation of Unactivated Alkenes With Molecular Oxygen | Catalysis | ChemRxiv | Cambridge Open Engage

Nickel‐Catalyzed Allylic Substitution Reactions: An Evolving Alternative - Ghorai - 2022 - European Journal of Inorganic Chemistry - Wiley Online Library
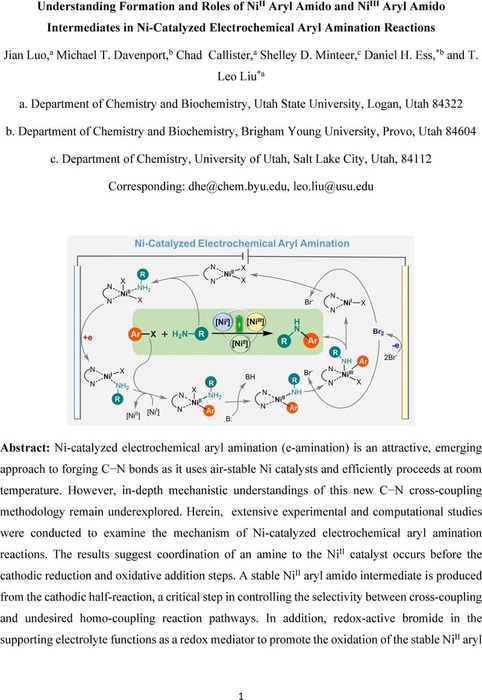
Understanding Formation and Roles of Ni(II) Aryl Amido and Ni(III) Aryl Amido Intermediates in Ni-Catalyzed Electrochemical Aryl Amination Reactions | Catalysis | ChemRxiv | Cambridge Open Engage

Synthesis and catalytic activity of nickel(II) complexes containing NCN pincer ligands - ScienceDirect

Radical Mechanism of IrIII/NiII-Metallaphotoredox-Catalyzed C(sp3)–H Functionalization Triggered by Proton-Coupled Electron Transfer: Theoretical Insight | CCS Chem

Proposed catalytic cycle for the nickel-catalyzed arylation of ethers... | Download Scientific Diagram
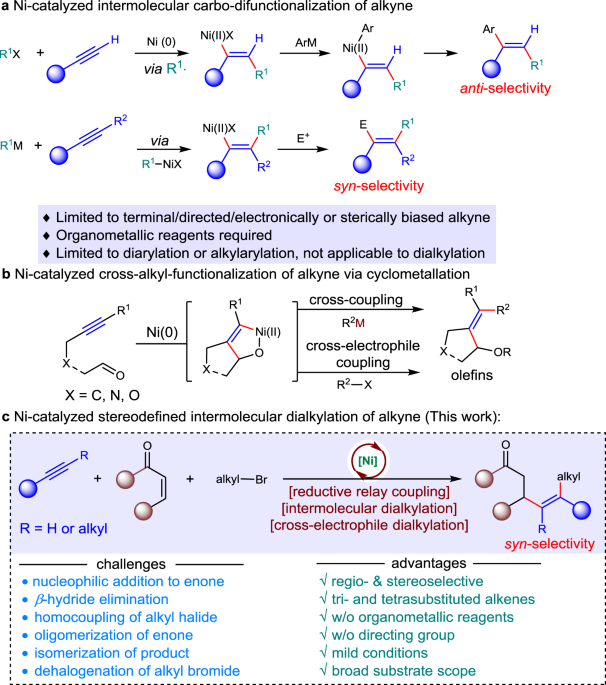
Ni-catalyzed regio- and stereo-defined intermolecular cross-electrophile dialkylation of alkynes without directing group | Nature Communications

DFT insight into mechanism of the Ni(II)-catalyzed enantioselective Michael addition: A combined computational and experimental study - ScienceDirect

Merging photoredox with nickel catalysis: Coupling of α-carboxyl sp3-carbons with aryl halides | Science
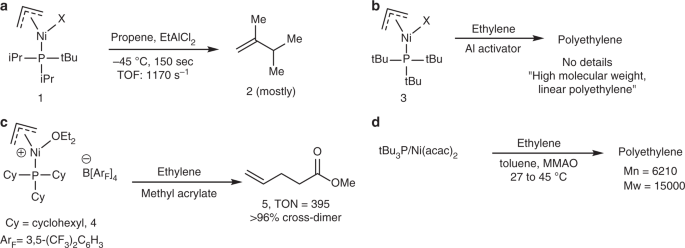
A highly active Ni(II)-triadamantylphosphine catalyst for ultrahigh-molecular-weight polyethylene synthesis | Nature Communications


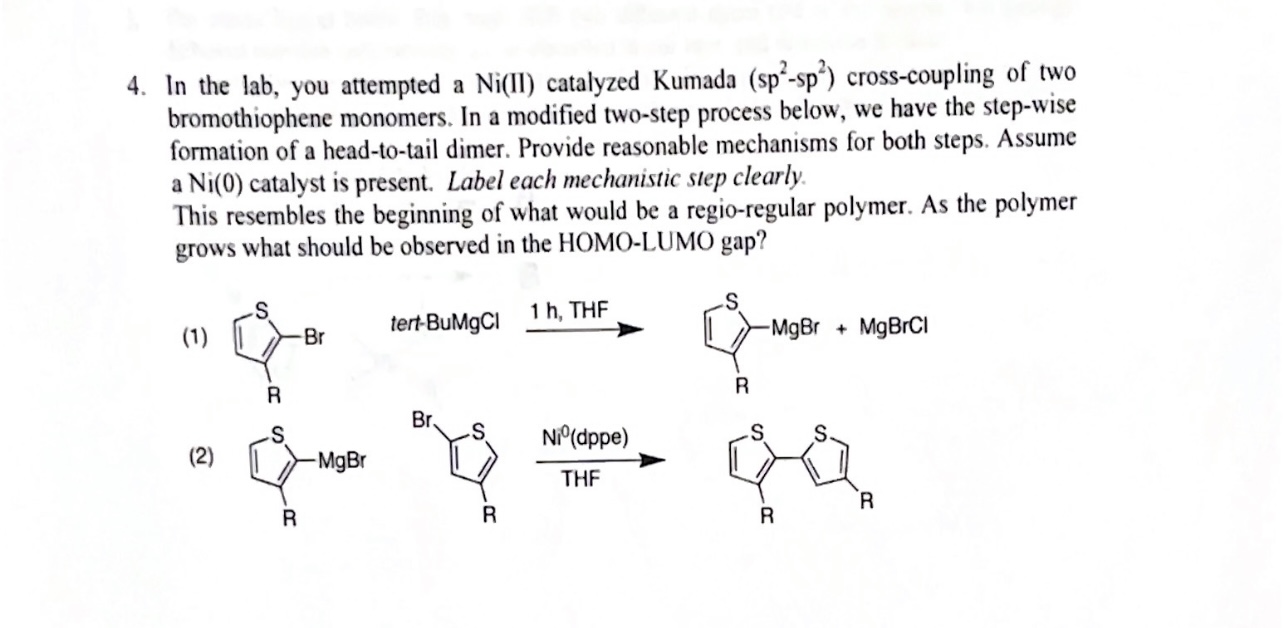
![ASAP] Nickel(II)–N?N?O Pincer Type Complex-Catalyzed N-alkylation o ASAP] Nickel(II)–N?N?O Pincer Type Complex-Catalyzed N-alkylation o](https://www.researcher-app.com/image/eyJ1cmkiOiJodHRwczovL3MzLWV1LXdlc3QtMS5hbWF6b25hd3MuY29tL3N0YWNrYWRlbWljL3Byb2R1Y3Rpb24vcGFwZXIvMzg3MDA0NC5wbmciLCJmb3JtYXQiOiJ3ZWJwIiwicXVhbGl0eSI6MTAwLCJub0NhY2hlIjp0cnVlfQ==.webp)
